Infra-Red Spectrometry
- Infra-red(IR) spectrometry uses the same principles as all spectrometry.
- It absorbs light at a specific wavelength and it is possible to detect this absorbance and identify the molecule.
- IR differs from UV by the light it absorbs. Infra-Red radiation is the light of main interest.
- IR radiation causes molecules to vibrate.
- The vibration of the bonds in a molecule depend on the energy absorbed.
- The IR spectrometer detects how the absorption varies with each bond and produces an IR spectrum.
IR Spectrometer
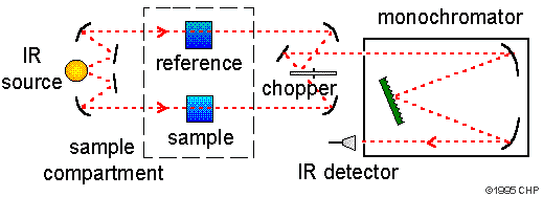
- The IR lamp emits light across the IR range.
- The light passes through the sample and the reference sample.
- The light is then passed through a chopper and a monochromator.
- This allows the correct light through to the detector.
- The computer translates the information and outputs and IR spectrum.
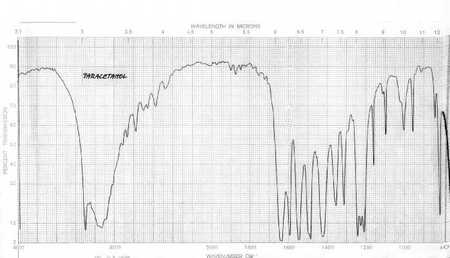
IR Spectrum
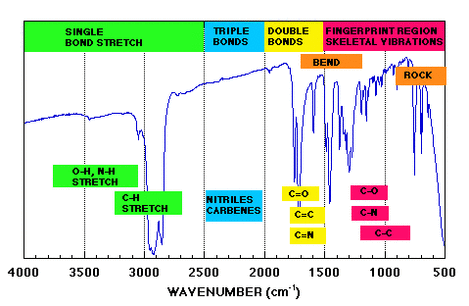
IR Spectrum
- IR Spectra are difficult to interpret as each type of bond in a compound will give a different peak.
- The diagram to the right shows a basic spectrum with some of the main areas where bonds will give peaks.
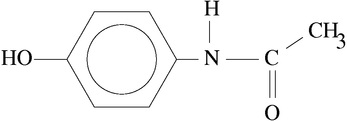
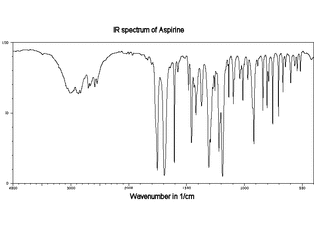
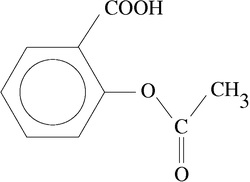
Comparison of Two Spectra
Uses of Infra-red Spectrometry
- IR is commonly used for structural determination.
- IR is being used to help identify the structure of complex molecules in space.
- Across the world many Breathalysers used by police contain IR spectrometers.