Atomic Absorption Spectrometry
- On of the hardest things as a chemist is that no matter how long you stare at a compound, you will never be able to tell exactly what it exactly is.
- Spectrometry can help with this. Molecules respond to light energy when they are hit. This response can be measured and information on the molecule can be obtained.
- Spectrometry is the study of the way light and matter interact.
- There are a number of spectroscopic techniques and a basic principle is shared by all.
- This principle is:
Light
- Light carries energy of tiny particles called photons.
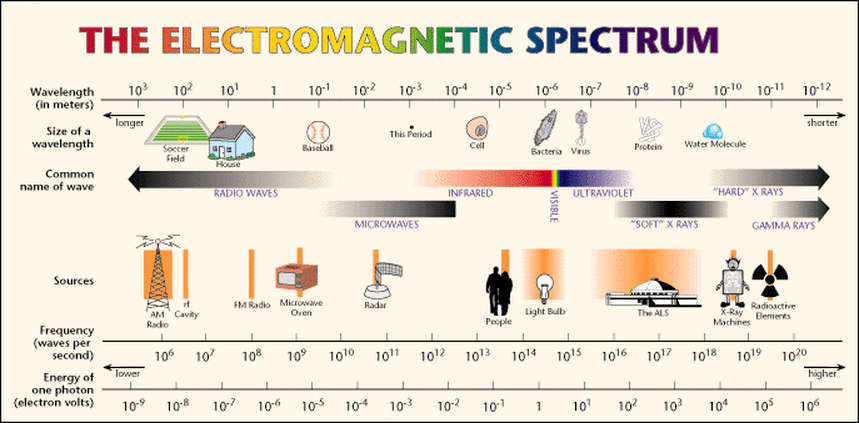
- Light has wave properties with characteristic wavelengths and frequency.(see above diagram)
- The energy of the photon is related to the frequency and the wavelength.
- The range of wavelengths and frequencies in light is known as the electromagnetic spectrum..
- The spectrum is divided into various regions from short wavelengths to long wavelengths.
- The lower the wavelength the higher the energy. Examples of high energy waves are X-Rays and Gamma Rays.
- The higher the wavelength the lower the energy. Examples of low energy waves are Microwaves and Radio waves.
- The visible region is the light we can see. It is only a small section of the spectrum.
Principle of Spectrometry
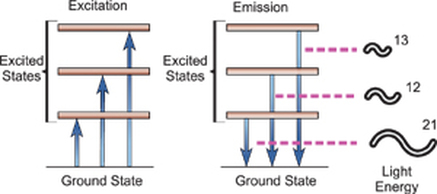
- When matter absorbs electromagnetic radiation the change which occurs depends on the amount of energy being absorbed.
- Absorption of energy causes an electron to jump from the ground state to a high energy state.
- This change in energy allows scientists to learn more about the chemical and physical properties of the molecule.
Types of Spectrometry
There are various different types of Spectrometry which can be used to identify molecules. Some of these are:
- Ultraviolet-Visible Spectrometry
- Infra-Red Spectrometry
- Mass Spectrometry
- Flame Atomic Absorption Spectrometry
- Graphite Furnace Atomic Absorption Spectrometry.