Thin Layer Chromatography
- Like all chromatographic techniques TLC requires a mobile phase and a stationary phase.
- the stationary phase is usually a metal, plastic or glass plate coated with a thin layer of solid.
- The most common solids used in TLC are aluminium oxide or silica gel. They are mixed with a binder such as calcium sulphate.
- The mobile phase used in TLC is a solvent which is suitable to separate the chemical mixture.
Sample Application
- Sample application is a critical step in achieving good separation in TLC.
- The following method is the ideal procedure for applying the sample spot to the paper:
- Draw a line using a ruler and pencil approximately 1 cm from the edge of the plate.
- Use a capillary tube to add a spot of the sample to the line. The spot should have a diameter as small as possible.
- After the first spot is applied it leave it to dry. The sample should be applied to the same spot three to four time depending on how dilute the sample is.
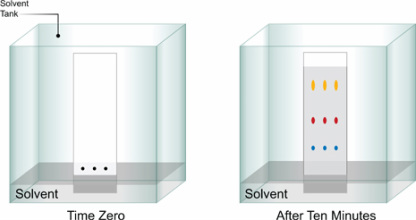
Plate Development
- Plate development is the next stage in TLC.
- Plate development is the process by which the sample is carried up the plate by the mobile phase.
- The plate is place in a closed saturated container containing the mobile phase.
- The mobile phase is drawn up the plate capillary action and once it passes the line where the sample was applied it dissolves the samples and carries them up the stationary phase.
- Once the mobile phase has reached approximately two thirds up the plate, it is removed and left to dry.
- Sometimes it is not possible to see the spots on the plate. There are a few methods to locate them.
- One method is to spray iodine on the plate. The iodine reacts with the organic compounds to yield a dark product which is visible on the plate.
- Often a fluorescent material is added to the plate which under UV light will display the spots.
Retention Factor (Rf)
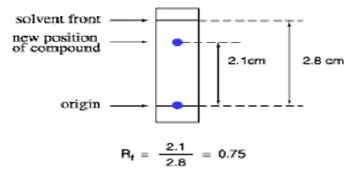
Rf value Calculation
- The Retention Factor is the distance travelled by the spot divided by the solvent.
- The Rf value can provide corroborative evidence to help identify a compound.
- If two compounds have the same Rf value then they are likely to be the same compound.
Uses of Thin Layer Chromatography

- TLC is currently the most commonly used planar chromatographic method used today.
- It is faster, has better resolution and is more sensitive than paper chromatography.
- It has become widely used in the forensic and drug industry.
- In the drug industry it can be used to detect the purity of drugs and can often be used in the identification of
organic compounds. - In forensic science TLC is a useful technique in detecting chemicals. Some of these chemicals include:
- Chemical Weapons,
- Explosives,
- Gun Powder,
- Illicit Drugs.
- TLC can also be used to separate dyes taken from fabrics found at the scene of a crime.